Dans le monde à enjeux élevés de la biotechnologie et de la fabrication pharmaceutique, la frontière entre un produit mondial réussi et un cauchemar réglementaire est souvent tracée par la qualité des machines sur le plancher de l'usine. Alors que nous avançons en 2026, la complexité des molécules biologiques — allant des protéines de recombinaison aux peptides bioactifs avancés — nécessite un niveau de contrôle environnemental que les équipements industriels traditionnels ne peuvent tout simplement pas fournir. C'est pourquoi une machine hautement spécialiséeRéacteur en acier inoxydableest devenu la pierre angulaire de la production moderne, garantissant le respect strict deNormes des équipements biopharmaceutiquespour toute installation.
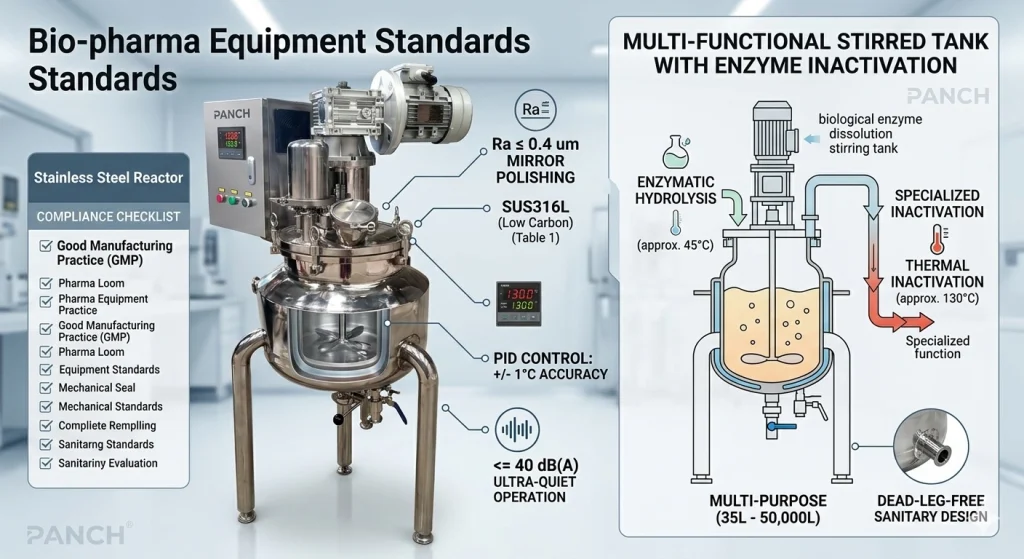
Pour les ingénieurs, les responsables du contrôle qualité et les spécialistes des achats, la compréhension de ces protocoles ne consiste plus seulement à cocher une case pour un auditeur local. Il s'agit de garantir la pérennité d'une installation face aux réglementations évolutives, d'assurer la cohérence d'un lot à l'autre et de protéger des investissements de plusieurs millions de dollars par le biais de démarches rigoureusesBonnes pratiques de fabrication (BPF).
Dans le secteur pharmaceutique, le choix de l'acier inoxydable constitue la première ligne de défense contre l'échec d'un lot. Pour se conformer àNormes des équipements biopharmaceutiques, le matériau doit résister à la corrosion chimique et aux cycles de stérilisation à haute température sans libérer d'ions métalliques dans le produit.
| Fonctionnalité | Acier industriel standard (SUS304) | Degré bio-pharmaceutique (SUS316L) | Impact de la conformité |
| Résistance à la corrosion | Modéré | Supérieur (Molybdène élevé) | Prévient la formation de piqûres lors de réactions acides |
| Finition de surface (Ra) | <= 0,8 μm | <= 0,4 μm (Polissage miroir) | Essentiel pourBonnes pratiques de fabrication (BPF) |
| Contenu en carbone | <= 0,08 % | <= 0,03 % (Basse Empreinte Carbone) | Prévient la dégradation de la soudure à 130°C |
| Facilité de nettoyage CIP | Efficacité de 85 % | Efficacité de 100 % | Pas de zones mortes pour la croissance bactérienne |
Aperçu d'expert :La rugosité de la surface n'est pas seulement une question esthétique. Dans la bio-pharmaceutique, un Ra de 0,4 μm constitue la « ligne de sécurité biologique ». Tout matériau plus rugueux permet aux biofilms de se fixer, rendant la stérilisation standard inefficace.
Une question récurrente dans « Les gens demandent aussi » est :Pourquoi la rugosité de la surface est-elle importante en pharmacie ?De haut niveauNormes des équipements biopharmaceutiquesspécifier une poli miroir avec une rugosité deRa ≤ 0,4 μmpour prévenir l'adhérence microbienne, en garantissant que chaque cycle de pulvérisation à 360 degrés soit efficace à 100 % et prêt pour l'audit.
Dans le contexte d'unRéacteur en acier inoxydableLe décalage thermique peut être fatal à l'activité biologique. Que vous réalisiez une hydrolyse enzymatique ou une inactivation thermique, la capacité à contrôler la chaleur est un pilier essentiel de l'ingénierie moderne.
Les éléments de chauffage traditionnels marche/arrêt causent un « dépassement de température », où le matériau continue de chauffer même après l'arrêt de l'alimentation. Pour répondre aux exigences modernesNormes des équipements biopharmaceutiques, nos réacteurs utilisentContrôleurs intelligents PID (Proportionnel-Intégral-Dérivé).
Lorsqu'il est associé à une haute sensibilitéCapteurs Pt100, ces systèmes atteignent une précision de± 1 °CImaginez un processus nécessitant l'inactivation d'une enzyme : le système doit rapidement passer d'une température de réaction stable de 45°C à une température létale de 130°C et la maintenir précisément. Toute déviation pendant cette courbe pourrait conduire àdénaturation thermiquedes mauvaises protéines. En maintenant ces repères internes stricts, les fabricants s'alignent surBonnes pratiques de fabrication (BPF)pour assurer l'efficacité biologique.
L'un des sujets les plus discutés sur Quora concernant la conception d'usine est « La fatigue de l'opérateur ». Historiquement, le plateau pharmaceutique était une cacophonie de bruits de engrenages. Cependant, à l'échelle internationaleNormes des équipements biopharmaceutiquesOnt évolué pour inclure l'élément humain comme une priorité pour l'excellence opérationnelle.
| Type d'équipement | Niveau de bruit (décibels) | Évaluation de la sécurité au travail | Adéquation à la biopharma |
| Mélangeur d'engrenages standard | 75 – 85 dB(A) | Avertissement : Fatigue élevée | Pas recommandé pour les salles blanches |
| Agitateur industriel | 60 – 70 dB(A) | Modéré | Niveau alimentaire de base |
| Réacteur Bio-pharma Elite | <= 40 dB(A) | Optimal : Ultra-silencieux | Dépasse les normes des équipements biopharmaceutiques |
En mettant en œuvre des systèmes de transmission haute précision, notre équipement maintient un niveau de bruit de charge de<= 40 dB(A). Cette attention portée à « l'Ingénierie centrée sur l'humain » devient rapidement une partie obligatoire au sein de l'entrepriseNormes des équipements biopharmaceutiquespour les entreprises visant des opérations en salle blanche à haute efficacité.
Une tendance mise en avant par les analystes de l'industrie sur LinkedIn est la « Intensification des processus ». ModerneNormes des équipements biopharmaceutiquesencourager l'utilisation de récipients multifonctionnels afin de minimiser les risques de transfert et l'erreur humaine.
NotreBioréacteur de dissolution enzymatique biologique avec agitationest un exemple parfait. Elle intègre un mélangeur haute efficacité, un chauffage/refroidissement à chemise, et une résistance sous vide dans une seule unité. En regroupant ces fonctions pour répondre à tousBonnes pratiques de fabrication (BPF)directives, les entreprises réduisent le nombre de vannes et de joints d'étanchéité—les principaux points de défaillance etcontamination croiséedans n'importe quelle configuration bio-pharmaceutique.
Le nettoyage en place (CIP) et la stérilisation en place (SIP) sont les deux moteurs de la sécurité pharmaceutique. Tout récipient qui prétend répondre aux normes internationalesNormes des équipements biopharmaceutiquesdoit être conçu sans « coins morts ».
Cela signifie que chaque soudure doit être lisse, chaque vanne doit être de qualité sanitaire, et la sortie de décharge doit être située au point le plus bas. Cette conception « sans coude mort » est une pierre angulaire de laBonnes pratiques de fabrication (BPF)nécessaire pour les médicaments de grade injectable, garantissant qu'aucun résidu d'une précédente série ne puisse compromettre la suivante.
Alors que certains responsables des achats se concentrent uniquement sur le prix initial, les investisseurs avertis prennent en compte le Coût Total de Possession (CTP). Un équipement qui ne répond pas aux normes du secteur entraîne des pertes catastrophiques pour les lots. En investissant dans un réacteur d'élite qui dépasse les standards actuelsBonnes pratiques de fabrication (BPF), vous achetez une assurance pour la réputation de votre marque.
Q : En quoi les normes d’équipement biopharmaceutique diffèrent-elles entre les réacteurs de qualité alimentaire et ceux de qualité pharmaceutique ?
A:Les protocoles de qualité pharmaceutique exigent une traçabilité du matériau bien plus élevée (SUS316L) et un polissage plus strict (Ra ≤ 0,4 μm) pour garantir une exclusion complète des microbes, alors que les normes de qualité alimentaire tolèrent des tolérances plus élevées.
Q : Le niveau de bruit fait-il vraiment partie des normes internationales pour l'équipement ?
A:Bien que cela ne soit pas toujours une loi statutaire, des niveaux de bruit ultra-faibles (<= 40 dB(A)) sont désormais intégrés dans l'interneNormes des équipements biopharmaceutiquesdes principales entreprises de biotechnologie pour protéger la santé des opérateurs et la précision dans les environnements stériles.
Q : Comment votre équipement garantit-il une précision à long terme ?
A:Nous utilisons des contrôleurs PID avancés calibrés pour maintenir une précision de± 1 °CCe rigorisme technique garantit que chaque lot reste cohérent, répondant aux contrôles de qualité les plus exigeants etNormes des équipements biopharmaceutiquesdans le monde entier.